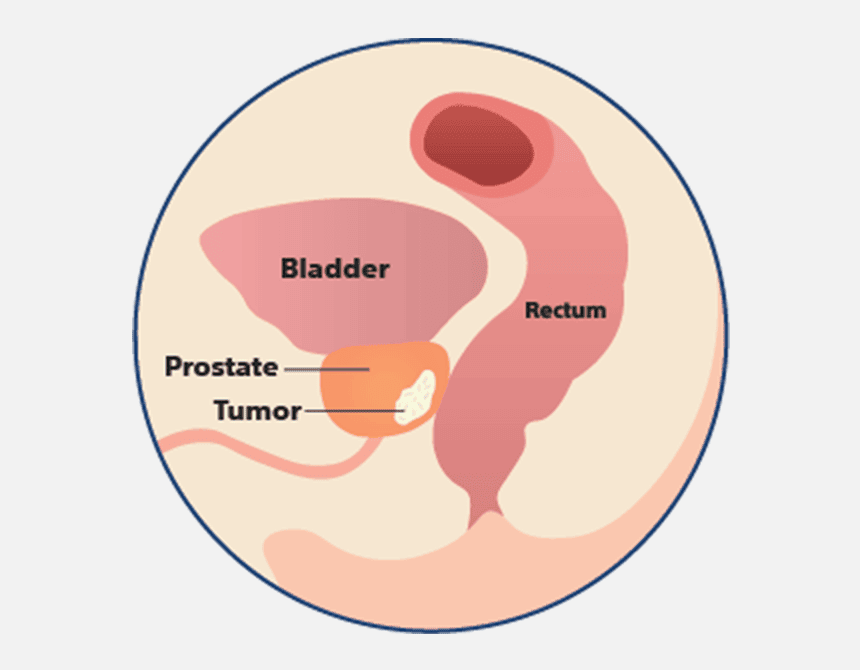
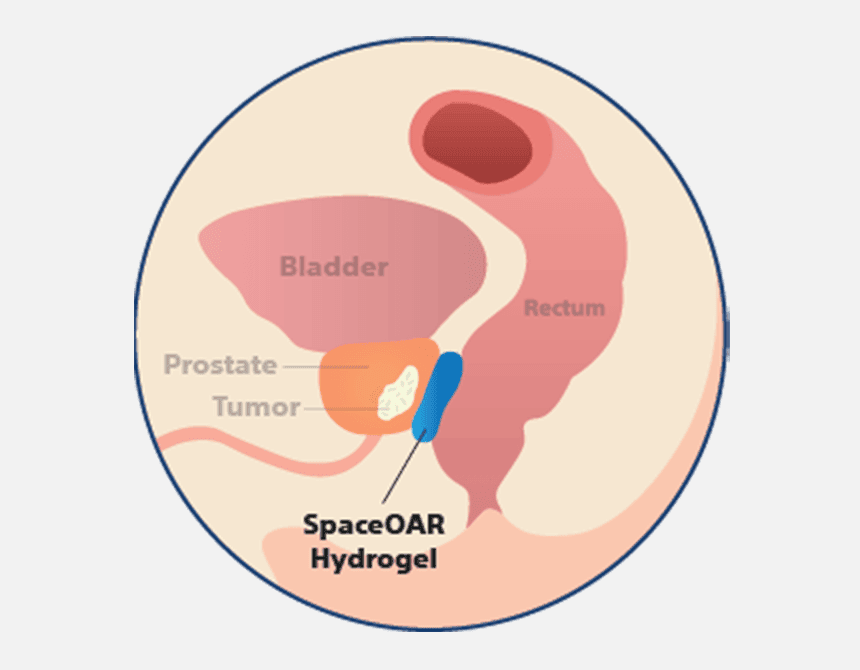
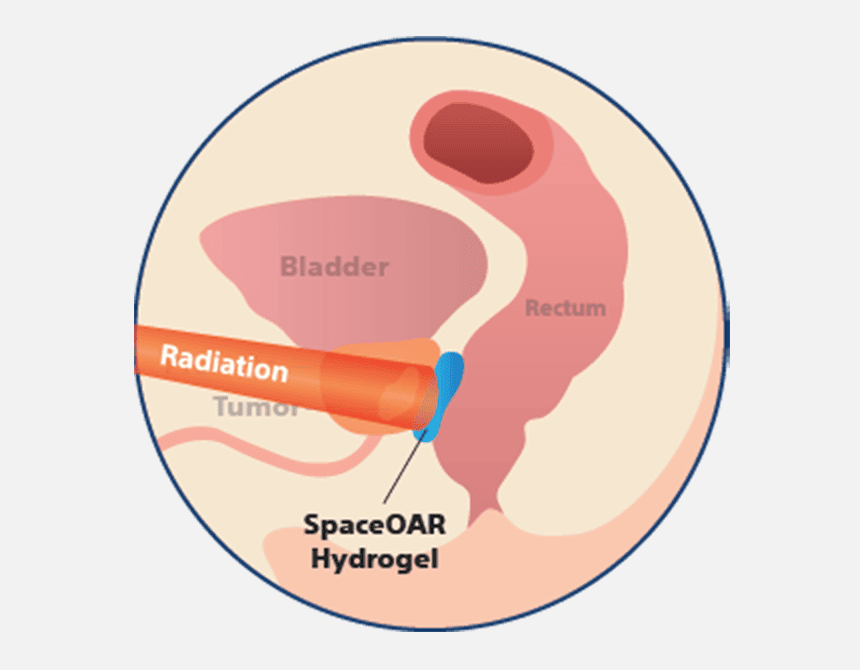
How does SpaceOAR™ Hydrogel work?
When treating prostate cancer patients with radiation therapy, the goal is to destroy the cancer cells while avoiding damage to surrounding healthy tissue. SpaceOAR Hydrogel, a polyethylene glycol (PEG) based hydrogel, is designed to help reduce the radiation dose delivered to the rectum during prostate cancer radiation treatments.1–3
What is the hydrogel made of?
SpaceOAR Hydrogel consists of mostly water (90%) and polyethylene glycol (PEG) that when combined, form a soft gel material. PEG materials have undergone extensive biocompatibility and safety testing and is used in a number of other medical applications such as surgical sealants used in the eye, brain, and spine.4
Have more questions? We can help.
Sign up to receive more information about SpaceOAR Hydrogel. Our patient education team can provide you with details about how SpaceOAR Hydrogel works, what to ask your doctor and how to find a doctor in your area.
Are you a physician looking for more information? Please click here.
References
- Mariados N, Sylvester J, Shah D, et al. Hydrogel spacer prospective multicenter randomized controlled pivotal trial: Dosimetric and clinical effects of perirectal spacer application in men undergoing prostate image guided intensity modulated radiation therapy. Int J Radiat Oncol Biol Phys. 2015 Aug 1;92(5):971–7.
- Hamstra DA, Mariados N, Sylvester J, et al. Continued benefit to rectal separation for prostate radiation therapy: Final results of a phase III trial. Int J Radiat Oncol Biol Phys. 2017 Apr 1;97(5):976–85.
- Hamstra D, Shah D, Kurtzman S, et al. Evaluation of sexual function on a randomized trial of a prostate rectal spacer. J Clin Oncol. 2017 February 20;35(Suppl 6):69.
- Data on file with Boston Scientific
Caution: U.S. Federal law restricts this device to sale by or on the order of a physician.
Results from case studies are not necessarily predictive of results in other cases. Results in other cases may vary.
All images are the property of Boston Scientific.